Infant Ibuprofen Recalled Due To A Possibility It Could Cause Permanent Kidney Damage
Recalls of various products happen all the time. And while they can be annoying (like when our kids’ favorite snack is recalled, or you know, ALL THE LETTUCE IN THE COUNTRY), it’s comforting to know that there are quality and safety measures in place to keep us safe. The latest safety recall has some parents freaking out, and for good reason. Liquid infant ibuprofen sold at three major retailers is being recalled. The items in question may contain higher concentrations of ibuprofen than is safe for infants to consume. For infants who are more susceptible to the drug, too much ibuprofen can lead to permanent kidney damage. This is definitely one recall you don’t want to take lightly, parents.
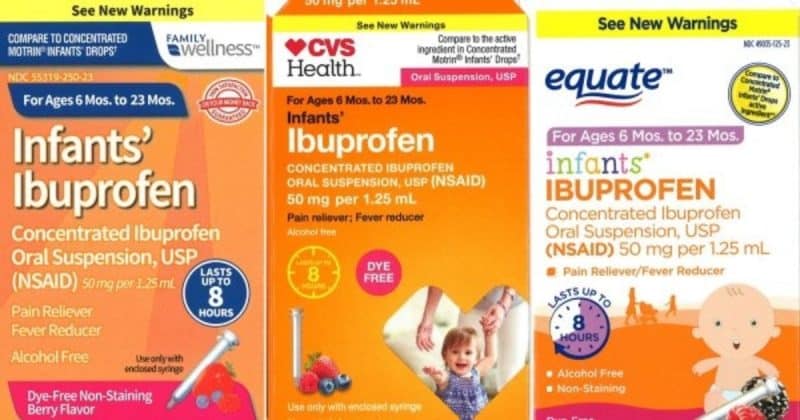
The recalled liquid infant ibuprofen is sold at CVS, Walmart, and Family Dollar.
Liquid Infant Ibuprofen Recalled Due to a Possibility It Could Cause Permanent Kidney Damage https://t.co/7QxUZnvb8G
— People (@people) December 7, 2018
Tris Pharma, which makes the over-the-counter drug, issued a voluntary recall for three lots of Infants’ Ibuprofen Concentrated Oral Suspension. The recalled medicines may have higher concentrations of ibuprofen per dose than is medically safe for an infant to consume. According to Tris Pharma, “Adverse effects that may be experienced are nausea, vomiting, epigastric pain, or more rarely, diarrhea. Tinnitus, headache and gastrointestinal bleeding are also possible adverse effects.”
The three lots being recalled are sold at CVS, Walmart, and Family Dollar stores around the country, under different names.
At Walmart stores, the medicine is sold under Equate Infants’ Ibuprofen Concentrated Oral Suspension,USP (NSAID), 50 mg per 1.25 mL,0.5 oz. bottle, with expiration dates of February 2019, April 2019 and August 2019. The lot numbers for the Equate liquid infant ibuprofen are 00717009A, 00717015A, and 00717024A. At CVS, the medicine is sold under Infants’ Ibuprofen Concentrated Oral Suspension, with an expiration date of August 2019, and lot number 00717024A. The liquid infant ibuprofen is sold at Family Dollar as Infants’ Ibuprofen Concentrated Oral Suspension, with the expiration date of August 2019 and lot number 00717024A.
So far, Tris Pharma has not received any reports of infants becoming sick after taking the medicine. If you have any of these meds at home, return them to the store where you bought them. For questions or concerns, you can contact Tris Pharma directly at 732-940-0358, Monday through Friday, 8:00am ET- 5:00pm PT. You can also email them directly here.





